Newborn Screening Notices
DSHS Laboratory Services Section – Notice to Public
Newborn Screening GAMT Result Reporting Statements
Texas Department of State Health Services (DSHS) Public Health Laboratory will begin screening for guanidinoacetate methyltransferase (GAMT) deficiency soon. As a result, newborn screening mailers may include one of nine new GAMT deficiency result reporting statements. Providers can review the full list of all possible results, analytes, and reporting notes by visiting the Newborn Screening – Healthcare Provider Resources webpage.
Questions?
Contact DSHS Newborn Screening Laboratory:
Phone: 512-776-7585
Email Newborn Screening: NewbornScreeningLab@dshs.texas.gov
Posted May 28, 2026
DSHS Newborn Screening Laboratory Closed May 25, 2026
The Texas Department of State Health Services (DSHS) Newborn Screening Laboratory will close on Monday, May 25, 2026, to observe Memorial Day.
Testing and reporting will resume on Tuesday, May 26, 2026.
To ensure specimens arrive at the lab promptly:
- DO NOT DELAY collection or shipment of newborn screening specimens.
- Ship dried specimens within 24 hours of collection. If mail or courier services are unavailable, ship as soon as possible.
- DSHS recommends shipping specimens using an overnight or trackable service like USPS priority mail, FedEx, and UPS.
For Questions Please Contact:
Newborn Screening Laboratory
Phone: 512-776-7585
Email: Newbornscreeninglab@dshs.texas.gov
Posted May 21, 2026
BEACONS-NBS Webinar for Health Care Professionals
The Association of Public Health Laboratories (APHL) will host an informational webinar on BEACONS-NBS, an NIH funded study exploring the feasibility of integrating genomic newborn screening into public health programs. The intended audience for this webinar is health care professionals.
What you will learn:
- The goals of BEACONS-NBS
- What results providers may see (rare, early onset, actionable)
- Workflow for follow-up and referrals
- How to access clinical resources and support
- Q&A session to follow the presentation
Who should attend?
- Pediatricians
- Primary care professionals
- OB/GYN/MFM providers
- Neonatal providers
- Geneticists and genetic counselors
- Medical specialists
- Midwives and doulas
- Public health professionals
- Other interested health care professionals
Speakers:
- Ingrid Holm, MD, MPH, Professor of Pediatrics, Harvard Medical School
- Melissa Wasserstein, MD, Professor of Pediatrics and Genetics, Albert Eistein College of Medicine
When?
Wednesday, June 3, 2026, 5-5:30pm PT / 8-8:30pm ET
Need help?
Contact DSHS Newborn Screening Laboratory:
Phone: 512-776-7585
Email Newborn Screening: NewbornScreeningLab@dshs.texas.gov
Posted May 20, 2026
Exchange Expired Insurance/Self-Pay Newborn Screening Collection Kits
Newborn Screening (NBS) collection kits with serial numbers that begin with 21 (21-XXXXXXX) have expired. Do not use these NBS collection kits. The Laboratory will reject NBS kits with serial numbers starting with 21 (21-XXXXXXX) if the date of collection occurs after March 31, 2026. If a specimen is rejected, a recollection will be required.
What you need to do now
- Check your inventory for NBS kits with serial numbers 21 (21-XXXXXXX).
- Remove all expired NBS kits from use immediately.
How to Exchange Insurance/Self-Pay NBS Collection Kits
- Complete the G-6D “Order Form for Newborn Screening Supplies (January 2026)”
- Count the number of expired Insurance/Self-Pay NBS kits with serial numbers that begin with 21 (21-XXXXXXX).
- Enter the number of expired Insurance/Self-Pay NBS kits in the “Quantity Requested” box.
- Write “Exchange” in the “Billing-Purchase Order Number” box.
Ship the completed form and expired Insurance/Self-Pay NBS kits to the Container Preparation Group using one of the addresses below:
Overnight/Courier Shipping
(FedEx, UPS, or other)
DSHS-Public Health Laboratory, MC 1947
Attn: Container Preparation Group
1100 West 49th Street
Austin, TX 78756-3199
USPS Regular and Priority Mail
(Regular Mail Service)
DSHS-Public Health Laboratory, MC 1947
Attn: Container Preparation Group
P.O. Box 149341
Austin, TX 78714-9341- The submitter is responsible for all shipping costs associated with returning expired NBS kits.
- Do not include requests for additional kits (see instructions below) on the same form used for exchanges. Container Preparation Group will process only the exchange from an exchange request.
- Submit exchange requests within three (3) months of the NBS kit expiration date. Container Preparation Group must receive exchange requests no later than June 30, 2026.
- Allow five (5) business days from receipt for Container Preparation Group to process exchange requests.
Important Reminder
- Do not return expired Medicaid/CHIP/Charity NBS collection kits. Discard them in the regular garbage, recycle them with paper, or shred them.
Ordering Additional Newborn Screening Collection Kit
- Complete a separate G-6D “Order Form for Newborn Screening Supplies (January 2026)” to order additional kits.
- Fax or email the completed form to the Container Preparation Group.
Questions?
Container Preparation Group
Phone: 512-776-7661
Fax: 512-776-7672
Email: containerprepgroup@dshs.texas.gov
Posted May 18, 2026
Newborn Screening Result Report Update: Addition of Guanidinoacetate Methyltransferase (GAMT) Deficiency
To prepare for the upcoming addition of GAMT deficiency to the Texas Newborn Screening (NBS) panel, we are providing an example of the updated disorder table from the NBS result report. The results for GAMT deficiency will appear at the bottom of the disorder table.
The visual guide will help facilities update or modify their reporting systems before the GAMT deficiency screening launches in June 2026.
We will distribute finalized result reporting statements prior to the implementation date.
Disorder | Screening Result |
| Amino Acid Disorders | Normal |
| Fatty Acid Disorders | Normal |
| Organic Acid Disorders | Normal |
| Galactosemia | Normal |
| Biotinidase Deficiency | Normal |
| Hypothyroidism | Normal |
| CAH | Normal |
| Hemoglobinopathies | Normal |
| Cystic Fibrosis | Normal |
| SCID | Normal |
| X-ALD | Normal |
| SMA | Normal |
| Lysosomal Diseases | Normal |
| GAMT Deficiency | Abnormal: See Note 1 |
Have questions?
Contact DSHS Newborn Screening Laboratory:
Phone: 512-776-7585
Email Newborn Screening: NewbornScreeningLab@dshs.texas.gov
Posted May 15, 2026
Updates to the 2026 Newborn Screening Collection Kits
The Texas Department of State Health Services (DSHS) Laboratory will start shipping the 2026 Newborn Screening (NBS) Collection Kits next week. The new kits will have serial numbers starting with 26 (26XXXXXXX).
What’s New in the 2026 NBS Collection Kits:
- Replaced “Uninsured” with “Unfunded” on all pages of the Medicaid/CHIP/Unfunded Kits (NBS 3). Unfunded applies to patients without insurance or self-pay who do not qualify for coverage of NBS services through Medicaid, CHIP, or any other government program. Use this type of NBS Collection Kit for newborns eligible for Medicaid, covered by CHIP (Children’s Health Insurance Program), or without any other payment options as required in Texas Administrative Code, Title 25 section 37.55. Use “unfunded” for families who do not have insurance, cannot afford to pay out of pocket, and do not qualify for any government programs. This represents an exception rather than standard practice.
The “Feed” field on the Demographic Form (page 3) has changed to “On TPN?”
Report TPN (Total Parenteral Nutrition) status because it can significantly affect newborn screening results, even if the baby received TPN for a short time. Mark “Y” in the box if the baby received TPN at any time before or during specimen collection. Leave the field blank if the baby did not receive TPN.

Revised the Texas Newborn Screening Instructions (page 6).
We revised the instructions to improve clarity and readability. The updated page is easier to read, less crowded, and includes website links for additional information. We also improved the instructions on the protective flap to make them clearer and easier to follow.
What Providers Should Do Now:
- Continue using your current NBS Collection Kits until they expire.
- Prepare the staff responsible for NBS collection for these upcoming changes so your team is ready when the new NBS Collection Kits arrive.
Questions?
Contact DSHS Newborn Screening Laboratory:
Phone: 512-776-7585
Email: NewbornScreeningLab@dshs.texas.gov
Posted May 14, 2026
The Newborn Screening Laboratory Will Soon Begin Screening for GAMT Deficiency
The Texas Department of State Health Services Texas Newborn Screening Program will soon begin screening for guanidinoacetate methyltransferase (GAMT) deficiency.
What to know:
- GAMT deficiency can cause serious neurological problems.
- GAMT deficiency requires early diagnosis and prompt treatment to prevent serious health complications.
- GAMT deficiency is a very rare condition.
Where can I find more information on GAMT deficiency?
- DSHS will share updates through email notices during implementation. Visit Baby’s First Test for additional information.
Will these tests require more blood?
The amount of blood we currently ask providers to collect is enough to run all tests. Make sure ALL pre-printed circles on the filter paper are completely filled.
Specimen Collection Resources:
- Specimen Quality Improvement | Texas DSHS
- Making a Difference Through Newborn Screening: Blood Collection on Filter Paper (NBS01-Ed7).
Have questions?
Contact DSHS Newborn Screening Laboratory:
Phone: 512-776-7585
Email Newborn Screening: NewbornScreeningLab@dshs.texas.gov
Posted May 11, 2026
Exchange Expired Insurance/Self-Pay Newborn Screening Collection Kits
Newborn Screening (NBS) collection kits with serial numbers that begin with 21 (21-XXXXXXX) have expired. Do not use these NBS collection kits. The Laboratory will reject NBS kits with serial numbers starting with 21 (21-XXXXXXX) if the date of collection occurs after March 31, 2026. If a specimen is rejected, a recollection will be required.
What you need to do now
- Check your inventory for NBS kits with serial numbers 21 (21-XXXXXXX).
- Remove all expired NBS kits from use immediately.
How to Exchange Insurance/Self-Pay NBS Collection Kits
- Complete the G-6D “Order Form for Newborn Screening Supplies (January 2026)”
- Count the number of expired Insurance/Self-Pay NBS kits with serial numbers that begin with 21 (21-XXXXXXX).
- Enter the number of expired Insurance/Self-Pay NBS kits in the “Quantity Requested” box.
- Write “Exchange” in the “Billing-Purchase Order Number” box.
Ship the completed form and expired Insurance/Self-Pay NBS kits to the Container Preparation Group using one of the addresses below:
Overnight/Courier Shipping
(FedEx, UPS, or other)
DSHS-Public Health Laboratory, MC 1947
Attn: Container Preparation Group
1100 West 49th Street
Austin, TX 78756-3199
USPS Regular and Priority Mail
(Regular Mail Service)
DSHS-Public Health Laboratory, MC 1947
Attn: Container Preparation Group
P.O. Box 149341
Austin, TX 78714-9341- The submitter is responsible for all shipping costs associated with returning expired NBS kits.
- Do not include requests for additional kits (see instructions below) on the same form used for exchanges. Container Preparation Group will process only the exchange from an exchange request.
- Submit exchange requests within three (3) months of the NBS kit expiration date. Container Preparation Group must receive exchange requests no later than June 30, 2026.
- Allow five (5) business days from receipt for Container Preparation Group to process exchange requests.
Important Reminder
- Do not return expired Medicaid/CHIP/Charity NBS collection kits. Discard them in the regular garbage, recycle them with paper, or shred them.
Ordering Additional Newborn Screening Collection Kit
- Complete a separate G-6D “Order Form for Newborn Screening Supplies (January 2026)” to order additional kits.
- Fax or email the completed form to the Container Preparation Group.
Questions?
Container Preparation Group
Phone: 512-776-7661
Fax: 512-776-7672
Email: containerprepgroup@dshs.texas.gov
Posted April 1, 2026
DSHS Newborn Screening Laboratory Closure on March 7, 2026
Due to ongoing Laboratory Renovations, the Texas Department of State Health Services (DSHS) Newborn Screening Laboratory will not be open on March 7, 2026.
To ensure specimens arrive at the lab promptly:
- DO NOT DELAY collection or shipment of newborn screening specimens.
- Ship dried specimens within 24 hours of collection. If mail or courier services are unavailable, ship as soon as possible.
- DSHS recommends shipping specimens using an overnight or trackable service like USPS priority mail, FedEx, and UPS.
For Questions Please Contact:
Newborn Screening Laboratory
Phone: 512-776-7585
Email: Newbornscreeninglab@dshs.texas.gov
Posted March 5, 2026
Ongoing Newborn Screening Laboratory Renovations
Over the weekend of February 28, 2026, the DSHS Laboratory building will be having renovation work done. Due to this, result reporting may experience a one-day delay.
Full testing and reporting will resume on Monday, March 2, 2026.
Continue to ensure specimens arrive at the lab promptly:
- DO NOT DELAY collection or shipment of newborn screening specimens.
- Ship dried specimens within 24 hours of collection. If mail or courier services are unavailable, ship as soon as possible.
- DSHS recommends shipping specimens using an overnight or trackable service like USPS priority mail, FedEx, and UPS.
For Questions Please Contact:
Newborn Screening Laboratory
Phone: 512-776-7585
Email: Newbornscreeninglab@dshs.texas.gov
Posted February 27, 2026
Newborn Screening Quality Improvement Hints: Documenting Gestational Age
Accurate information on the Newborn Screening demographic form is vital for testing and result follow-up. Gestational age is a crucial measure used for assessing the baby’s health at birth. Document the gestational age in weeks and days at birth. If unknown, the specimen remains acceptable.
Download a copy of the Quality Improvement Hint: Documenting Gestational Age
Texas DSHS Newborn Screening Laboratory
Phone: 512-776-7585
Email: NewbornScreeningLab@dshs.texas.gov
Posted February 19, 2026
Newborn Screening Collection Kits Expiring March 31, 2026
Newborn Screening (NBS) Collection Kits with serial numbers that begin with 21 (21-XXXXXXX) will expire on March 31, 2026.

Please take the following actions:
- Check your current NBS kit inventory.
- Identify any NBS kits with serial numbers starting with 21 (21-XXXXXXX).
- Collection of specimens on the NBS kits with serial numbers starting with 21 (21-XXXXXXX) must occur on or before March 31, 2026.
After March 31, 2026:
- The Laboratory will reject NBS kits with serial numbers starting with 21 (21-XXXXXXX) if the date of collection occurs after March 31, 2026. If the Laboratory rejects a specimen, it will require a recollection.
- You may exchange NBS kits labeled Insurance/Self-Pay. You cannot return them for a refund or credit. We will send more information about the exchange program after the expiration date, March 31, 2026.
- You should discard NBS kits labeled Medicaid/CHIP/Charity in the regular garbage, recycle them with paper, or shred them.
For Questions Please Contact:
Newborn Screening Laboratory
Phone: 512-776-7585
Email: Newbornscreeninglab@dshs.texas.gov
Posted February 17, 2026
DSHS Newborn Screening Laboratory Closed February 16, 2026
The Texas Department of State Health Services (DSHS) Newborn Screening Laboratory will close Monday, February 16, 2026, to observe Presidents’ Day.
Testing and reporting will resume on Tuesday, February 17, 2026.
To ensure specimens arrive at the lab promptly:
- DO NOT DELAY collection or shipment of newborn screening specimens.
- Ship dried specimens within 24 hours of collection. If mail or courier services are unavailable, ship as soon as possible.
- DSHS recommends shipping specimens using an overnight or trackable service like USPS priority mail, FedEx, and UPS.
For Questions Please Contact:
Newborn Screening Laboratory
Phone: 512-776-7585
Email: Newbornscreeninglab@dshs.texas.gov
Posted February 13, 2026
Newborn Screening Laboratory – Delayed Start for January 27
Due to continued severe weather and driving conditions, the Texas Newborn Screening Laboratory will open at 12:00PM for testing on Tuesday, January 27, 2026.
- Expect delays in patient result reporting.
- Please continue shipping all specimens according to standard guidelines.
- Result reporting, including out-of-range results, will experience delays. Staff will complete reporting as soon as possible.
- DSHS Clinical Care Coordination will provide follow-up outreach services once results become available.
Providers should continue to:
- Act immediately if an infant shows signs or symptoms of a potential critical newborn screening condition.
- Recognize that newborn screening results may not yet be available.
- Initiate diagnostic evaluation or treatment as clinically indicated.
- Contact Newborn Screening Clinical Care Coordination for assistance with follow-up recommendations and referral to appropriate specialist.
- Expedite follow-up activities for out-of-range results whenever possible.
- Ensure providers document, communicate, and discuss results with the family or caregiver.
- Facilitate repeat or confirmatory testing, specialty referral, and timely intervention as needed.
- Collect and dry newborn screens within the required timeframes.
- Collect the 1st screen at 24 - 48 hours of age, or before hospital discharge.
- Collect the 2nd screen between 7-14 days of age, a minimum of 168 hours.
- Ship specimens using normal delivery methods as soon as possible.
- Expect courier services to operate as usual but anticipate weather-related delays.
- Monitor courier service alerts for local updates.
- Store dried specimens at room temperature in a dry location during courier delays.
- Do not place specimens in airtight or sealed containers.
While navigating severe weather, the DSHS Newborn Screening Program appreciates your continued commitment to protecting the health of Texas newborns.
Questions?
Newborn Screening Laboratory:
Phone: 512-776-7585
Email: NewbornScreeningLab@dshs.texas.gov
Clinical Care Coordination Emergency Contact:
Phone: 512-939-1216
Email: Newborn@dshs.texas.gov
Posted January 25, 2026
2026 Newborn Screening Laboratory Closure
Due to severe weather conditions, Texas Newborn Screening Laboratory will be closed for testing on Monday, January 26, 2026.
- The laboratory plans to resume testing on Tuesday, January 27, 2026, pending weather conditions.
- Please continue shipping all specimens according to standard guidelines.
- Result reporting, including out-of-range results, will experience delays. Staff will complete reporting as soon as possible.
- DSHS Clinical Care Coordination will provide follow-up outreach services once results become available.
Providers should continue to:
- Act immediately if an infant shows signs or symptoms of a potential critical newborn screening condition.
- Recognize that newborn screening results may not yet be available.
- Initiate diagnostic evaluation or treatment as clinically indicated.
- Contact Newborn Screening Clinical Care Coordination for assistance with follow-up recommendations and referral to appropriate specialist.
- Expedite follow-up activities for out-of-range results whenever possible.
- Ensure providers document, communicate, and discuss results with the family or caregiver.
- Facilitate repeat or confirmatory testing, specialty referral, and timely intervention as needed.
- Collect and dry newborn screens within the required timeframes.
- Collect the 1st screen at 24 - 48 hours of age, or before hospital discharge.
- Collect the 2nd screen between 7-14 days of age, a minimum of 168 hours.
- Ship specimens using normal delivery methods as soon as possible.
- Expect courier services to operate as usual but anticipate weather-related delays.
- Monitor courier service alerts for local updates.
- Store dried specimens at room temperature in a dry location during courier delays.
- Do not place specimens in airtight or sealed containers.
While navigating severe weather, the DSHS Newborn Screening Program appreciates your continued commitment to protecting the health of Texas newborns.
Questions?
Newborn Screening Laboratory:
Phone: 512-776-7585
Email: NewbornScreeningLab@dshs.texas.gov
Clinical Care Coordination Emergency Contact:
Phone: 512-939-1216
Email: Newborn@dshs.texas.gov
Posted January 25, 2026
Newborn Screening Guidance During Severe Weather
As you prepare for potential severe weather, please remember the following:
- Do Not Delay Newborn Screening (NBS) Specimen Collection.
- Collect 1st screens at 24-48 hours of age.
- Collect 2nd screens between 7-14 days of age, a minimum of 168 hours.
- Verify parent or guardian contact information and ensure it remains current in case of relocation.
- For patient transfers:
- Document newborn screening specimen collection status in transfer paperwork.
- Notify receiving facility if staff did not collect the NBS specimen before transfer.
- Allow NBS specimens to dry for a minimum of 3 hours.
- DO NOT place specimens in air-tight or sealed containers.
- Ship as soon as possible.
- If you use courier services, contact the courier for location-specific guidance.
- During courier delays, store dried specimens at room temperature in a dry location.
- Walk your specimens inside the postal location.
- Do not drop place specimens in outdoor blue mailboxes.
- Mailboxes could become humid and can flood during high water events.
Ensuring specimens arrive at the laboratory in good condition helps support accurate newborn screening results.
Need help?
Contact DSHS Newborn Screening Laboratory:
Phone: 512-776-7585
Email: NewbornScreeningLab@dshs.texas.gov
Posted January 23, 2026
DSHS Newborn Screening Laboratory Closure
Scheduled for January 10, 2026
The DSHS Newborn Screening Laboratory will not perform testing on Saturday, January 10, 2026, due to laboratory maintenance.
Routine newborn screening (NBS) testing will not occur on this day.
The clinical care coordination registered nurses will report time critical disorder results to providers on Saturday, January 10, 2026.
Testing and reporting will resume on Monday, January 12, 2026.
To ensure specimens arrive at the lab promptly:
- DO NOT DELAY collection or shipment of newborn screening specimens.
- Ship dried specimens within 24 hours of collection. If mail or courier services are unavailable, ship as soon as possible.
- DSHS recommends shipping specimens using an overnight or trackable service like USPS priority mail, FedEx, and UPS.
For Questions Please Contact:
Newborn Screening Laboratory
Phone: 512-776-7585
Email: Newbornscreeninglab@dshs.texas.gov
Posted January 09, 2026
2026 Is Here
Incorrect dates DELAY specimen testing and can cause a specimen to be UNSATISFACTORY for testing.
To Avoid Delays:
- Ensure dates (including year) are complete and accurate.
- Assign staff to double-check all specimen information is:
- Complete
- Accurate
- Legible
- Respond to The Department of State Health Services (DSHS) request for clarifying information.
- Specimens are unsatisfactory for testing if a response containing requested information is not received by 1:00 p.m. the next business day.

Need help?
Contact DSHS Newborn Screening Laboratory:
Phone: 512-776-7585
Email Newborn Screening: NewbornScreeningLab@dshs.texas.gov
Posted January 02, 2026
Updated Order form for Newborn Screening Supplies
Effective January 1, 2026, the Department of State Health Services (DSHS), Public Health Laboratory revised the newborn screening (NBS) testing fees to support the addition of new conditions, increased operational costs, and expanded courier services.
Please use the current G-6D Newborn Screening Supply Order Form (revision date: 01/2026) when placing orders. You can find the updated form and the Laboratory Fee Schedule on the Laboratory Testing Services Manual - Forms and Laboratory Fee Schedule | Texas DSHS webpage.
We cannot process orders submitted on outdated forms.
For Ordering Questions Contact:
Laboratory Supply
Phone: 512-776-7661
Email: containerprepgroup@dshs.texas.gov
Posted January 02, 2026
2026 Will Soon Be Here
Incorrect dates DELAY specimen testing and can cause a specimen to be UNSATISFACTORY for testing.
To Avoid Delays:
- Ensure dates (including year) are complete and accurate.
- Assign staff to double-check all specimen information is:
- Complete
- Accurate
- Legible
- Respond to The Department of State Health Services (DSHS) request for clarifying information.
- Specimens are unsatisfactory for testing if a response containing requested information is not received by 1:00 p.m. the next business day.

Need help?
Contact DSHS Newborn Screening Laboratory:
Phone: 512-776-7585
Email Newborn Screening: NewbornScreeningLab@dshs.texas.gov
Posted December 30, 2025
Newborn Screening End-of-the-Year Ordering
Effective January 1, 2026, the Department of State Health Services (DSHS), Public Health Laboratory is revising newborn screening (NBS) testing fees to support the addition of new conditions, increased operational costs, and expanded courier services. The ordering system will update on January 1, 2026, to capture these new costs.
- Effective January 1, 2026: The cost of Insurance/Self-Pay NBS kits will increase from $68.63 to $94.81.
- We will bill all Insurance/Self-Pay NBS kits ordered on or after this date at the new price of $94.81.
Place orders now:
- Submit your orders before 12:00 PM (noon) on Wednesday, December 31, 2025, to receive current pricing.
- Orders received after 12:00 PM December 31, 2025, will not get processed, and will require resubmission on the updated G-6D Newborn Screening Supply Order Form.
- The updated G-6D Newborn Screening Supply Order Form will be available Friday, January 2, 2026.
We cannot approve overrides for orders received after the deadline of 12:00 PM (noon), December 31, 2025.
For Ordering Questions Contact:
Laboratory Supply
Phone: 512-776-7661
Email: containerprepgroup@dshs.texas.gov
Posted December 22, 2025
2026 Will Soon Be Here
Incorrect dates DELAY specimen testing and can cause a specimen to be UNSATISFACTORY for testing.
To Avoid Delays:
- Ensure dates (including year) are complete and accurate.
- Assign staff to double-check all specimen information is:
- Complete
- Accurate
- Legible
- Respond to The Department of State Health Services (DSHS) request for clarifying information.
- Specimens are unsatisfactory for testing if a response containing requested information is not received by 1:00 p.m. the next business day.

Need help?
Contact DSHS Newborn Screening Laboratory:
Phone: 512-776-7585
Email Newborn Screening: NewbornScreeningLab@dshs.texas.gov
Posted December 18, 2025
Update on Newborn Screening For
Duchenne Muscular Dystrophy
The 89th Texas Legislature passed a bill which authorizes the Texas Newborn Screening Program to test for Duchenne Muscular Dystrophy (DMD), including follow-up services. We cannot begin DMD testing at this time because the current Texas Department of State Health Services (DSHS) Laboratory does not have space for the required equipment.
The Legislature approved funding (capital authority) for DSHS to use federal funds to build the additional laboratory space. The project to build the additional laboratory space began this year and will take about five years to complete. Once the additional space is complete, DSHS will validate and implement DMD testing and follow-up services.
We will share updates as progress continues. Thank you for supporting Texas newborns.
For Questions Please Contact:
Newborn Screening Laboratory
Phone: 512-776-7585
Fax: 512-776-7157
Email: newbornscreeninglab@dshs.texas.gov
Posted December 17, 2025
DSHS Newborn Screening Laboratory Holiday Closure
Christmas Closure: Wednesday, December 24, 2025, clinical care coordination registered nurses will report time critical disorder results to providers. Routine newborn screening (NBS) testing will not occur this day.
Thursday, December 25, 2025, the Texas Department of State Health Services (DSHS) NBS Laboratory will not be open, and no testing will occur.
Friday, December 26, 2025, the NBS Laboratory will resume all testing.
New Year’s Day Closure: Thursday, January 1, 2026, the NBS Laboratory will not be open, and no testing will occur.
DO NOT Delay Collection or Shipment
- Ship dried NBS specimens within 24 hours of collection. If mail or courier services are unavailable, ship as soon as possible.
- DSHS recommends shipping NBS specimens using an overnight or trackable service like USPS Priority Mail, FedEx, or UPS.
- If you use one of these services, plan accordingly for your site to ensure specimens are not delayed due to shipping.
Newborn Screening Supply Order Requests
- Standard orders received before noon Tuesday, 12/23/2025, will get processed and shipped via FedEx Ground.
- Priority overnight orders received by noon on Tuesday, 12/23/2025, will get processed and shipped via FedEx Priority Overnight.
- Orders received after noon Tuesday, 12/23/2025, through Friday, 12/26/2025, will get processed Monday, 12/29/2025.
- Orders received on Thursday, 1/1/2026, will get processed on Friday, 1/2/2026.
Need help?
Contact DSHS Newborn Screening Laboratory:
Phone: 512-776-7585
Email Newborn Screening: NewbornScreeningLab@dshs.texas.gov
Order Questions: ContainerPrepGroup@dshs.texas.gov
Posted December 15, 2025
Avoid Holiday Shipping Delays
Newborn screening (NBS) specimens are unsatisfactory if received after 13 days of collection.
TIPS TO AVOID SPECIMEN REJECTION DURING HOLIDAYS:
- Allow NBS specimens to dry a minimum of three hours, per Clinical and Laboratory Standards Institute (CLSI).
- Ship dried specimens same day using an overnight or trackable service.
- Keep in mind that courier and mail services may have delays during the holidays.
- DO NOT hold specimens for bulk mailing or shipping.
- Ensure all specimens have the correct name, date of birth, and date of collection before shipping. Incorrect or incomplete demographic information can lead to testing delays or rejection.
- Assign a staff person to double check all NBS specimens. Check for spot quality and all demographic information is complete before shipment.
- Ship to the correct address:
For Overnight/Courier Shipping (UPS, FedEx, other):
Texas Department of State Health Services
Public Health Laboratory Division, MC 1947
1100 W. 49th Street
Austin, TX 78756-3199
For USPS Regular and Priority Mail:
Texas Department of State Health Services
Public Health Laboratory Division, MC 1947
PO Box 149341
Austin, TX 78714-9341
Need help?
Contact DSHS Newborn Screening Laboratory
Phone: 512-776-7585
Email: NewbornScreeningLab@dshs.texas.gov
Posted December 08, 2025
DSHS Austin Newborn Screening Laboratory
Thanksgiving Holiday Closure
On Thursday, November 27, 2025, the Texas Department of State Health Services (DSHS) Austin Newborn Screening (NBS) Laboratory will not be open, and no testing will occur. Please see below for more details:
- The NBS Laboratory will resume testing on Friday, 11/28/2025.
- DO NOT delay collection or shipment of NBS specimens.
- Ship dried specimens within 24 hours of collection. If mail or courier services are unavailable, ship as soon as possible.
- DSHS recommends shipping specimens using an overnight or trackable service like USPS Priority Mail, FedEx, or UPS.
- If you use one of these services, plan accordingly for your site to ensure specimens are not delayed due to shipping.
Newborn Screening Supply Order Requests
- Standard orders received before noon Wednesday, 11/26/2025, will get processed and shipped via FedEx Ground.
- Priority overnight orders received on Wednesday, 11/26/2025, will get processed and shipped via FedEx Priority Overnight.
- Orders received after noon Wednesday, 11/26/2025, through Sunday, 11/30/2025, will get processed Monday, 12/01/2025.
Need help?
Contact DSHS Newborn Screening Laboratory
Phone: 512-776-7585
Email: NewbornScreeningLab@dshs.texas.gov
Container Preparation: ContainerPrepGroup@dshs.texas.gov
Posted November 17, 2025
Proposed Increase to DSHS Newborn Screening Kit Effective January 1, 2026
The Department of State Health Services (DSHS) Public Health Laboratory is revising newborn screening (NBS) testing and fees to support the addition of new conditions, increased operational costs, and expanded courier service.
Effective January 1, 2026:
- The cost of Insurance/Self-Pay NBS kits will increase from $68.63 to $94.81.
- We will bill all Insurance/Self-Pay NBS kits ordered on or after this date at the new price of $94.81.
- The procedure name on the DSHS Austin Laboratory Fee Schedule will update from “Newborn Screening Panel Test Kit (Private Pay) Fee” to “Newborn Screening Panel Test Kit (Insurance/Self-Pay) Fee.”
Insurance Reimbursement Guidance
To ensure your facility receives the appropriate reimbursement from insurance companies:
- Contact each health plan/insurance company to update your contracts with the new kit cost.
- Verify that the HCPCS code S3620 is correctly aligned with the new price of the NBS Collection Kit.
- Bill using HCPCS code S3620 for the full cost of the NBS Collection Kit. Do not bill separately for each condition on the panel.
Billing Notes
- Facilities may charge reasonable collection and processing fees in addition to the NBS Collection Kit cost.
- You may also bill using CPT code 36416 for collecting a capillary blood sample.
For the full DSHS Laboratory fee schedules, including current NBS fees, please visit: http://www.dshs.texas.gov/lab/fees.shtm
For Questions Please Contact:
DSHS Newborn Screening Laboratory
Phone: 512-776-7585
Email: NewbornScreeningLab@dshs.texas.gov
Posted November 06, 2025
Proposed increase to DSHS Newborn Screening Kit effective January 1, 2026
The Department of State Health Services (DSHS) Public Health Laboratory is revising laboratory testing and fees to meet business needs.
Effective January 1, 2026, the cost of paid newborn screening (NBS) kits increases from $68.63 to $94.81. Paid NBS kits, ordered on or after January 1, 2026, would be billed at the new price of $94.81. Additionally, the procedure name will be updated from “Newborn Screening Panel Test Kit (private pay) Fee” to “Newborn Screening Panel Test Kit (Insurance/Self-Pay) Fee.” The purpose of this increase will be for the addition of new conditions to the DSHS newborn screening panel and to support increased costs for operations.
Stakeholders may submit comments until December 31, 2025. To view complete details, visit the DSHS Laboratory’s Fee Schedule webpage.
Posted October 01, 2025
Newborn Screening Awareness Month:
The “Why” Behind What We Do
This month, we celebrate Newborn Screening (NBS) Awareness Month and 60 years of life saving screenings in Texas. We know why we do this - early detection saves lives.
We invite you to hear directly from families as they share their powerful stories. They speak about their journeys after diagnosis, how they manage their conditions, and the hope that they are not alone. Their voices remind us why every screen matters.
For the past 60 years, the Texas NBS Program has focused on early detection so babies can receive the care they need. We are proud of our past and excited for the future, and we look forward to the next 60 years protecting the smallest Texans.

Questions?
Contact DSHS Newborn Screening Laboratory:
Phone: 512-776-7585
Email: NewbornScreeningLab@dshs.texas.gov
Posted September 24, 2025
Newborn Screening Awareness Month:
Is Your Practice Up to Date?
As we celebrate Newborn Screening Awareness Month, it’s the perfect time to review your newborn screening (NBS) practices. An annual review helps ensure continued success and quality screening for patients. Ensure your team stays up to date with the latest guidance from the Texas NBS Program.
Annual NBS Review
Use the tools below to complete a quick self-review of your NBS process:
Review Your NBS Submitter Information
- Check that your facility name, address, phone number, and key contacts are up to date in the DSHS Laboratory Submitter system. You can do this by calling the Newborn Screening Educators at 888-963-7111, ext. 7585.
- If you need updates, visit the Forms webpage for the Submitter ID Number Request Form. Mark section 2 as “Updating Submitter Information”.
Complete the Newborn Screening Provider Self-Assessment
- Evaluate your NBS collection, submission, and follow-up practices.
- Access the Newborn Screening Provider Self-Assessment Checklist.
Request Staff Training (if needed)
- Based on your self-assessment results, determine if your staff would benefit from additional training.
- To request training, complete the DSHS Public Health Provider Training Request Form.
Your dedication makes a difference. Taking the time to review and refine your practices ensures the highest standard of care and helps us continue saving lives through early detection.
Questions
Contact DSHS Newborn Screening Laboratory:
Phone: 1-888-963-7111 ext. 7585 or Locally: 512-776-7585
Email Newborn Screening: NewbornScreeningLab@dshs.texas.gov
Posted September 17, 2025
Behind the Screen: Celebrating 60 Years of Teamwork
For 60 years, newborn screening in Texas has been more than a test, it has been a team effort. Every timely result depends on dedicated teams across the program:
- Specimen Check-In and Logistics: Receive, verify, and route newborn screening specimens for fast processing.
- Supply and Container Preparation: Provide Newborn Screening Kits and materials statewide to keep testing on track.
- Testing Teams: Perform lifesaving testing that identifies babies at risk for serious conditions.
- Informatics and Quality Assurance: Maintain accurate and secure data and processes for every screen.
- Education and Outreach: Train and support providers to collect high-quality specimens.
- Clinical Care Coordination: Ensures timely communication with providers after abnormal results so that babies receive fast treatment.
For six decades, these teams have advanced with science and technology to protect Texas babies.
Learn more about the people and processes behind newborn screening: We Are Newborn Screening | Texas DSHS.
Questions?
Contact DSHS Newborn Screening Laboratory:
Phone: 1-888-963-7111 ext. 7585 or Locally: 512-776-7585
Email Newborn Screening: NewbornScreeningLab@dshs.texas.gov
Posted September 10, 2025
60 YEARS OF NEWBORN SCREENING: THEN AND NOW
Since 1965, DSHS Newborn Screening (NBS) has continued to improve and expand newborn screening for babies born in Texas.
Let’s take a moment to reflect on how far we have come:
1965 | Today |
|---|---|
| Texas began newborn screening with a single condition: phenylketonuria (PKU), using dried blood spot testing. | Texas screens for 59 conditions using dried blood spots and performs two point-of-service screenings: hearing and critical congenital heart disease. |
| Pathology laboratories, the Texas Department of Health Laboratory, and hospitals performed PKU testing. | The Texas DSHS Public Health Laboratory in Austin performs all testing on newborn blood spots.
|
| All blood spot cards were handwritten and mailed in with limited tracking. | We work with facilities to promote electronic data entry and labeling. We encourage all providers to send their shipments using a trackable mail service. |
| Education for health care providers was by paper pamphlets. | Resources are available in real-time electronically for download. Training, webinars, guidelines, and support are available year-round. |
| Second screens were not performed. | Texas began requiring second screens collected at 7 to 14 days of age in 1983.
|
To learn more about the expansion of the Texas Newborn Screening program over the years, visit the Evolution of Newborn Screening in Texas.https://www.dshs.texas.gov/laboratory-services/programs-laboratories/newborn-screening-laboratory/we-are-newborn-screening
Join the 60-year celebration!
Consider sharing a story within your facility, hosting an educational moment with staff, or displaying a poster that highlights the importance of newborn screening.
Questions?
Contact DSHS Newborn Screening Laboratory: :
Phone: 1-888-963-7111 ext. 7585 or Locally: 512-776-7585
Email Newborn Screening: NewbornScreeningLab@dshs.texas.gov
Posted September 2, 2025
Happy 60th Birthday Texas Newborn Screening Program!
That’s right — we’re turning the BIG 6-0! For six incredible decades, the Texas Newborn Screening Program has been protecting tiny Texans, one heel prick at a time.
💙 60 years of life-changing early disease detection
💙 60 years of providing clinical care coordination for every baby affected
💙 60 years of protecting our littlest Texans
It all started in 1965 with one disorder — phenylketonuria, (PKU). Fast forward to today, and we are screening for over 50 conditions, each with available treatments that help give babies a better start in life.
This milestone is more than a number — it’s a celebration of laboratory science, health care, and teamwork.
So, grab some balloons, bake a cake, and throw your newborn screening program a birthday bash! Don’t forget to share the celebration and help us spread the word: newborn screening saves lives.
Here’s to 60 years of success — and to the bright future ahead!

For Questions Please Contact:
Phone: 1-888-963-7111 ext. 7585 or Local: 512-776-7585
Email Newborn Screening: NewbornScreeningLab@dshs.texas.gov
Posted August 27, 2025
Newborn Screening Laboratory Began Testing Lysosomal Diseases on August 18, 2025
The Texas Department of State Health Services added screening for Lysosomal Diseases to the Texas Newborn Screening Panel effective August 18, 2025.
Texas now screens for:
- Glycogen Storage Disease Type II (Pompe)
- Mucopolysaccharidosis Type I (MPS I)
- Mucopolysaccharidosis Type II (MPS II)
- Infantile Krabbe Disease (Krabbe)
What to Know:
- Pompe and Infantile Krabbe Disease are time critical disorders that require immediate action. Without prompt intervention, damage can begin in the first weeks of life.
- MPS I and MPS II are time sensitive disorders that demand prompt treatment to prevent serious health complications.
Where can I find more information on Lysosomal Diseases?
- DSHS Newborn Screening Program New Disorder Toolkit
- ACT sheets provide recommended immediate next steps for health care professionals.
- FACT sheets provide facts and information to parents.
- Baby’s First Test
Will these tests require more blood?
The amount of blood we currently ask providers to collect is enough to run all the tests. Make sure ALL pre-printed circles on the filter paper are completely filled.
Specimen Collection Resources:
- Specimen Quality Improvement | Texas DSHS
- Making a Difference Through Newborn Screening: Blood Collection on Filter Paper (NBS01-Ed7).
Will there be a fee increase?
DSHS plans to increase the fee on January 1, 2026, to include the cost of the new tests. DSHS will notify stakeholders at least 90 days in advance.
Need help?
Contact DSHS Newborn Screening Laboratory:
Phone: 1-888-963-7111 ext. 7585 or Local: 512-776-7585
Email Newborn Screening: NewbornScreeningLab@dshs.texas.gov
Posted August 18, 2025
Newborn Screening Lysosomal Disease Testing Result Reporting Statements **Revised**
The screening result note for Lysosomal Disease testing, possible mucopolysaccharidosis type II (Hunter syndrome) has changed. This is an update to the released result reporting statements on August 13, 2025.
The screening result note now reads

The change is also reflected on the Mucopolysaccharidoses Type 2 (MPS II) FACT Sheet for parents.
A full list of all possible results, analytes and notes for Lysosomal Diseases are currently available. Remember to always read screening result notes fully before acting.
Need help?
Contact DSHS Newborn Screening Laboratory:
Phone: 1-888-963-7111 ext. 7585 or Local: 512-776-7585
Email Newborn Screening: NewbornScreeningLab@dshs.texas.gov
Posted August 14, 2025
Newborn Screening Lysosomal Disease Testing Result Reporting Statements
With the addition of Lysosomal Diseases to the Texas DSHS Newborn Screening (NBS) Panel, mailers may now include one of 19 new Lysosomal Disease result reporting statements or one of two new partially unsatisfactory result notes. Testing will begin later this summer, but a full list of all possible results, analytes and notes for Lysosomal Diseases are currently available.
Examples of the updated result reports are on the Newborn Screening – Healthcare Provider Resources page.
Remember to always read screening result notes fully before acting.
Need help?
Contact DSHS Newborn Screening Laboratory:
Phone: 1-888- 963-7111 ext. 7585 or Locally: 512-776-7585
Email Newborn Screening: NewbornScreeningLab@dshs.texas.gov
Posted August 12, 2025
Newborn Screening Result Report Update: Addition of Lysosomal Diseases
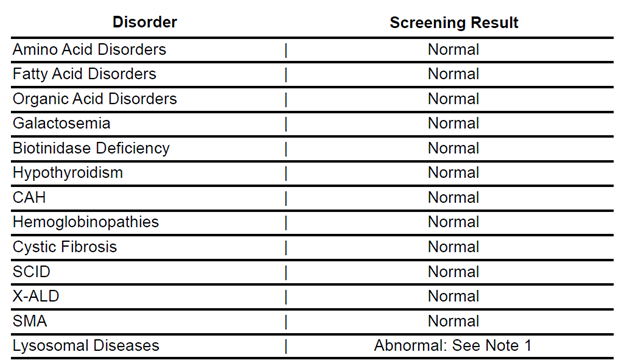
To prepare for the upcoming addition of Lysosomal Diseases to the Texas Newborn Screening (NBS) panel, we are providing an example of the updated disorder table from the NBS result report. The results for Lysosomal Diseases will appear at the bottom of the disorder table.
The visual guide will help facilities update or modify their reporting systems before the Lysosomal Disease screening launch later this summer.
We will distribute finalized result reporting statements prior to the implementation date.
Questions?
Contact DSHS Newborn Screening Laboratory:
Phone: 1-888- 963-7111 ext. 7585 or Locally: 512-776-7585
Email Newborn Screening: NewbornScreeningLab@dshs.texas.gov
Posted July 17, 2025
Updates to the 2025 Newborn Screening Collection Kits
The Texas Department of State Health Services (DSHS) Laboratory will start shipping the 2025 Newborn Screening (NBS) Collection Kits starting next week. The new kits will have serial numbers starting with 25 (25XXXXXXX).
What’s New in the 2025 NBS Collection Kits:
- Replaced “Charity” with “Uninsured” on all pages of the Medicaid/CHIP/Uninsured Kits (NBS 3). Uninsured applies to patients without insurance or self-pay who do not qualify for coverage of NBS services through Medicaid, CHIP, or any other government program. Use this type of NBS Collection Kit for newborns eligible for Medicaid, covered by CHIP (Children’s Health Insurance Program), or without any other payment options as required in Texas Administrative Code, Title 25 section 37.55.
- Changed the name of the “Parent Decision Form for Storage and Use of Newborn Screening Blood Spot Cards” to “Storage and Use of Newborn Screening Blood Spots” (page 2).
- Replaced “Ambiguous” with “Unknown” in the Sex field under the Newborn Information section on the Demographic form (page 3).
- Added “DO NOT COVER BARCODE” on the manila blood collection paper (page 5). This serves as a reminder to avoid placing labels over the barcodes.
- Revised the Texas Newborn Screening Instructions (page 6):
- Added “Apply only one blood drop to each circle” to Collection Procedure #4.
- Updated Collection Procedure #8 to say, “If possible, ship dried specimen the same day as collection.”
- Listed both the overnight courier address and USPS post office box side by side.
- Added a QR code linking to the CLSI blood collection video. (Note: The QR code no longer works. Providers can view the video directly at: https://youtu.be/S51Y9ShD6HI?si=hCdRoIZcCT7i9K2R)
- Added the Laboratory contact email to use when ordering more NBS Collection Kits.
What Providers Should Do Now:
- Continue using your current NBS Collection Kits until they expire.
- Prepare the staff responsible for NBS collection for these upcoming changes so your team is ready when the new NBS Collection Kits arrive.
Questions?
Contact DSHS Newborn Screening Laboratory:
Phone: 1-888- 963-7111 ext. 7585 or Locally: 512-776-7585
Email Newborn Screening: NewbornScreeningLab@dshs.texas.gov
Posted July 03, 2025
Texas Summer Weather Can Affect Specimen Quality
Shipping newborn screening (NBS) specimens during summer months can be challenging. High temperatures, humidity, and rain can compromise specimen quality and lead to inaccurate test results. Whenever possible, transport specimens under controlled conditions to protect their integrity.
Tips to help keep specimens safe during the summer:
- Schedule mail pickups inside your location. Do not place specimens on the outside of the facility door for pickup.
- Walk your specimens inside the postal location. Do not drop specimens in the blue mailboxes outside. Mailboxes become very hot and humid.
- Never leave specimens inside a non-running closed automobile.
Ensuring samples reach the lab in good condition helps provide accurate screening test results.
Need help?
Contact DSHS Newborn Screening Laboratory:
Phone: 1-888- 963-7111 ext. 7585 or Locally: 512-776-7585
Email Newborn Screening: NewbornScreeningLab@dshs.texas.gov
Posted July 01, 2025
Lysosomal Diseases Screening Coming Summer 2025
The Texas Newborn Screening Program will expand screening in Summer 2025 to include screening for Lysosomal Diseases.
Texas will begin screening for:
- Mucopolysaccharidosis Type I (MPS I)
- Mucopolysaccharidosis Type II (MPS II)
- Infantile Krabbe Disease (Krabbe)
- Glycogen Storage Disease Type II (Pompe)
What to Know:
- Pompe and Infantile Krabbe Disease are both time critical disorders. Time critical disorders are conditions where symptoms and potentially irreversible damage could develop in the first week of life.
- For all disorders, early recognition and treatment can reduce risk of morbidity and mortality.
Will these tests require more blood?
No, continue to completely fill ALL pre-printed circles on the filter paper.
Specimen Collection Resources:
- Specimen Quality Improvement | Texas DSHS
- Making a Difference Through Newborn Screening: Blood Collection on Filter Paper (NBS01-Ed7).
Will there be a fee increase?
DSHS plans to increase the fee on January 1, 2026, to include the cost of the new tests. DSHS will notify stakeholders at least 90 days in advance.
Where can I find more information on Lysosomal Diseases?
DSHS will share updates through email notices during implementation. Visit Baby’s First Test for additional information.
Have questions?
Contact DSHS Newborn Screening Laboratory:
Phone: 1-888-963-7111 ext. 7585 or locally: 512-776-7585
Email Newborn Screening: NewbornScreeningLab@dshs.texas.gov
Posted June 09, 2025
DSHS Laboratory Building Maintenance:
Reference Laboratory to Process Newborn Screening Specimens
The Texas Department of State Health Services (DSHS) Public Health Laboratory in Austin, Texas, is undergoing building updates to prevent unexpected shutdowns. This work will occur Friday, May 30 through Sunday, June 1, 2025.
To ensure timely newborn screening, DSHS has partnered with the reference laboratory Revvity Omics, to test a small portion of Texas Newborn Screening specimens. Revvity Omics meets regulatory testing requirements.
Key Updates for Submitters:
- Submitters will continue to receive result reports from DSHS except in a different format.
- Reports will include a cover page with demographics and submitter information.
- Attached to the cover page will be the reference laboratory report.
- Submitters will receive results via mail, fax, or web application as usual.
- Submitters do not need to contact the reference laboratory for results.
- Results will be available through the Texas Newborn Screening Web Application and Laboratory Reporting.
- DSHS Clinical Care Coordination will continue follow-up on screen-positive results.
Electronic (HL7) Ordering and Reporting:
- Normal Results: Reported via HL7, as usual.
- Non–normal Results:
- Will include an out-of-range/abnormal determination and an abnormal flag.
- The result note will refer to the physical report for details.
- A physical report, including reference laboratory results, will get mailed to the submitter and be available via the Texas NBS Web Application.
Newborn screening is time sensitive. Delayed submissions can impact infant health outcomes. Please continue sending specimens promptly to the Austin DSHS Public Health Laboratory to ensure timely testing and follow-up care.
Thank you for your dedication to newborn screening and the health of Texas newborns.
Need help?
Contact DSHS Newborn Screening Laboratory:
Phone: 1-888- 963-7111 ext. 7585 or Locally: 512-776-7585
Email Newborn Screening: NewbornScreeningLab@dshs.texas.gov
Posted May 27, 2025
Laboratory Closed May 26, 2025
The Texas Department of State Health Services Public Health Laboratory will close on May 26, 2025, to observe Memorial Day.
Need Help?
Contact the DSH Laboratory:
Phone: 1-888-963-7111
Visit: Laboratory Testing Services Manual - Specimen Receiving, Lab Hours, Holidays | Texas DSHS
Posted May 23, 2025
A Successful Newborn Screen Starts with a Quality Collection
Healthcare providers collecting the newborn screening heel stick blood specimen are the first step to a successful newborn screen. Completely filling all five circles on a newborn screening collection kit gives the state laboratory enough blood to test for all disorders.
When collecting a newborn screen, fill each circle, one at a time, until all five circles are completely filled with blood. Each circle of the filter paper holds a specific amount of blood. Take the time to check to make sure that the blood applied to the preprinted circle has soaked through to the other side. The other side should not have any white filter paper showing within the blood drop. Incomplete saturation of the filter paper or unfilled circles can cause the specimen to be unsatisfactory for all or some tests. When you have moved to the next circle for collection, do not go back to add more blood to previously filled circles. Adding more blood to the specimen can result in caked, clotted, or layered specimens, resulting in too much blood for proper testing.
For tips on collecting and improving the quality of your newborn screens please visit Specimen Quality Improvement | Texas DSHS. A video on how to perform blood collection on filter paper based off of CLSI documents is available here: Making a Difference Through Newborn Screening: Blood Collection on Filter Paper (NBS01-Ed7). Please share these collection tips with all staff that participate in specimen collections.
Have questions?
Contact DSHS Newborn Screening Laboratory:
Phone: 1-888- 963-7111 ext. 7585 or Locally: 512-776-7585
Email: NewbornScreeningLab@dshs.texas.gov
Posted April 29, 2025
United States Postal Service (USPS) Newborn Screening Specimen Shipping Issues
The Texas Department of State Health Services (DSHS) has received multiple reports from healthcare providers of USPS postal workers denying shipment of dried blood spots for newborn screening. These specimens are not subject to regulation under USPS Publication 52, Section 346.234d, meaning they are not regulated as Division 6.2 infectious substances, and you can mail them if they meet packaging requirements. USPS Packing Instructions 6G state that you must enclose the primary specimen in a secondary container.
Packaging Requirements:
- Allow specimens to dry for a minimum of three hours.
- Cover the specimen with the attached flap, which serves as the secondary container and bears the international biohazard symbol for USPS shipping regulations.
- Place the specimen in a blue diamond envelope provided by DSHS or a similar protective envelope. This envelope serves as the outer shipping container in compliance with USPS shipping regulations.
Recommended Actions:
- Inform USPS Staff: When mailing these specimens, provide a clear indication that the package contains dried blood spots as defined in USPS Publication 52, Section 346.234d and packaged according to USPS Packing Instruction 6G.
- Carry Documentation: It may be helpful to have a printed copy of this document or the relevant regulation to provide clarification if needed.
- Ask to speak to the Postmaster if issues persist.
The DSHS Laboratory is actively collaborating with USPS to resolve this matter and ensure efficient processing of your specimen shipments.
Need help?
Contact DSHS Newborn Screening Laboratory:
Phone: 1-888- 963-7111 ext. 7585 or Locally: 512-776-7585
Email: NewbornScreeningLab@dshs.texas.gov
Posted March 5, 2025
DSHS Laboratory Building Maintenance: Reference Laboratory to Process Newborn Screening Specimens
The Texas Department of State Health Services (DSHS) Public Health Laboratory in Austin, Texas, is undergoing building updates to prevent unexpected shutdowns. During this time, the Laboratory will have a series of planned weekend shutdowns.
To ensure timely newborn screening, DSHS has partnered with the reference laboratory Revvity Omics, which meets regulatory testing requirements.
Key Updates for Submitters:
- Submitters will continue to receive result reports from DSHS except in a different format.
- Reports will include a cover page with demographics and submitter information.
- Attached to the cover page will be the reference laboratory report.
- Submitters will receive results via mail, fax, or web application as usual.
- Submitters do not need to contact the reference laboratory for results.
- Results will be available through the Texas Newborn Screening Web Application.
- DSHS Clinical Care Coordination will continue follow-up on screen-positive results.
Electronic (HL7) Ordering and Reporting:
- Normal Results: Reported via HL7, as usual.
- Non–normal Results:
- Will include an out-of-range/abnormal determination and an abnormal flag.
- The result note will refer to the physical report for details.
- A physical report, including reference laboratory results, will get mailed to the submitter and be available via the Texas NBS Web Application.
Newborn screening is time sensitive. Delayed submissions can impact infant health outcomes. Please continue sending specimens promptly to ensure timely testing and follow-up care.
Thank you for your dedication to newborn screening and the health of Texas newborns.
Need help?
Contact DSHS Newborn Screening Laboratory:
Phone: 1-888- 963-7111 ext. 7585 or Locally: 512-776-7585
Email Newborn Screening: NewbornScreeningLab@dshs.texas.gov
Posted February 13, 2025
Newborn Screening Guidance During Inclement Weather
As you prepare for possible winter weather, please remember:
- Do Not Delay Newborn Screening (NBS) Specimen Collection.
- Collect 1st screens at 24-48 hours of age.
- Collect 2nd screens between 7-14 days of age, a minimum of 168 hours.
- Ensure the parent/guardian contact information will be valid throughout any potential relocation.
- For patient transfers - Include newborn screening specimen collection status in transfer documentation. Notify receiving facility of specimen collection need if not collected before transfer.
- Allow NBS specimens to dry a minimum of 3 hours.
- DO NOT put specimens in air-tight sealed containers.
- Ship As Soon As Possible.
- If you use courier services, contact the courier for extra information about your location.
- During courier delays store dried specimens at room temperature in a dry location.
- If using USPS, walk your specimens inside the postal location. Do not drop specimens in the blue mailboxes outside.
Ensuring samples reach the lab in good condition helps provide accurate screening test results.
Need help?
Contact DSHS Newborn Screening Laboratory:
Phone: 1-888- 963-7111 ext. 7585 or Locally: 512-776-7585
Email Newborn Screening: NewbornScreeningLab@dshs.texas.gov
Posted January 08, 2025
2025 Is Here
Incorrect dates DELAY specimen testing and can cause a specimen to be UNSATISFACTORY for testing.
To Avoid Delays:
- Ensure dates (including year) are complete and accurate.
- Assign staff to double-check all specimen information is:
- Complete
- Accurate
- Legible
- Respond to The Department of State Health Services (DSHS) request for clarifying information.
- Specimens are unsatisfactory for testing if information is not received by 1:00 p.m. the next business day.

Need help?
Contact DSHS Newborn Screening Laboratory:
Phone: 1-888- 963-7111 ext. 7585 or for local calls: 512-776-7585
Email Newborn Screening: NewbornScreeningLab@dshs.texas.gov
Posted January 02, 2025
Newborn Screening (NBS) for X-Linked Adrenoleukodystrophy (X-ALD) has Resumed Testing for all Specimens
With the resolution of instrument issues, DSHS has resumed testing X-ALD for all specimens.
- Regular testing for X-ALD has resumed.
- Revised reports with final X-ALD screening results are being sent for affected specimens originally reported as ‘Test Pending. Final report with results for this disorder to follow’.
- For submitters receiving electronic results, initial and revised results will transmit as usual for all affected specimens.
- For submitters who receive results by mail or by fax, you will receive your revised results by mail.
- Submitters who receive their results online will also get a copy by mail.
The DSHS Newborn Screening Program appreciates the dedication and commitment of healthcare providers to ensure the best possible outcomes for Texas newborns.
Questions?
Contact Newborn Screening Laboratory:
Phone: 1 (888) 963-7111 ext. 7333 or 512-776-7333
Email: NewbornScreeningLab@dshs.texas.gov
Posted December 23, 2024
DSHS Newborn Screening Laboratory Holiday Closure Reminder
Christmas Closure: Tuesday, December 24 and Wednesday, December 25, 2024, the Texas Department of State Health Services (DSHS) Newborn Screening (NBS) Laboratory will not be open, and no testing will occur. The NBS Laboratory will resume testing on Thursday, December 26, 2024.
New Year’s Day Closure: Wednesday, January 1, 2025, the Laboratory will not be open, and no testing will occur.
DO NOT Delay Collection or Shipment
- Ship dried NBS specimens within 24 hours of collection. If mail or courier services are unavailable, ship as soon as possible.
- DSHS recommends shipping NBS specimens using an overnight or trackable service like USPS Priority Mail, FedEx, or UPS.
- Per the FedEx website, FedEx will not be open on Wednesday, 12/25/2024 and Wednesday, 1/1/2025. FedEx will operate on a modified schedule December 23, 24, 26, 30 and 31. Please plan accordingly for your site.
Newborn Screening Supply Order Requests
- Standard orders received before noon Monday, 12/23/2024 will get processed and shipped via FedEx Ground.
- Priority overnight orders received by noon on Monday, 12/23/2024 will get processed and shipped via FedEx Priority Overnight.
- Orders received after noon Monday, 12/23/2024 through Thursday, 12/26/2024 will get filled Friday, 12/27/2024.
- Orders received on 1/1/2025 will get processed on 1/2/2025.
Need help?
Contact DSHS Newborn Screening Laboratory:
Phone: 1-888-963-7111 ext. 7585 or for local calls: 512-776-7585
Email Newborn Screening: NewbornScreeningLab@dshs.texas.gov
Container Preparation: ContainerPrepGroup@dshs.texas.gov
Posted December 19, 2024
Newborn Screening for X-Linked Adrenoleukodystrophy (X-ALD)
Due to unexpected issues with testing instrumentation, DSHS is temporarily unable to complete X-ALD testing for some specimens.
- Affected specimens for the X-ALD disorder will report as “Test Pending”. A result reporting note will state, “Final report with results for this disorder to follow”.
- When testing is complete, DSHS will send a revised report with a final X-ALD screening result. The result will be on a regular DSHS result report.
- For submitters receiving electronic results, initial and revised results will transmit as usual for all affected specimens.
- DSHS will provide more communication on this issue and notify submitters when regular testing and reporting of X-ALD has resumed.
The DSHS Newborn Screening Program appreciates the dedication and commitment of healthcare providers to ensure the best possible outcomes for Texas newborns.
Questions?
Contact Newborn Screening Laboratory:
Phone: 1 (888) 963-7111 ext. 7333 or Locally: 512-776-7333
Email: NewbornScreeningLab@dshs.texas.gov
Posted December 17, 2024
2025 Will Soon be Here
Incorrect dates DELAY specimen testing and can cause a specimen to be UNSATISFACTORY for testing.
To Avoid Delays:
- Ensure dates (including year) are complete and accurate.
- Assign staff to double-check all specimen information is:
- Complete
- Accurate
- Legible
- Respond to The Department of State Health Services (DSHS) request for clarifying information.
- Specimens are unsatisfactory for testing if information is not received by 1:00 p.m. the next business day.

Need help?
Contact DSHS Newborn Screening Laboratory:
Phone: 1-888- 963-7111 ext. 7585 or for local calls: 512-776-7585
Email Newborn Screening: NewbornScreeningLab@dshs.texas.gov
Posted December 16, 2024
Avoid Holiday Shipping Delays
Newborn screening (NBS) specimens are unsatisfactory if received after 13 days of collection.
TIPS TO AVOID SPECIMEN REJECTION DURING HOLIDAYS:
- Allow NBS specimens to dry a minimum of three hours, per Clinical and Laboratory Standards Institute (CLSI).
- Ship dried specimens same day using an overnight or trackable service.
- Keep in mind that courier and mail services may have delays during the holidays.
- DO NOT hold specimens for bulk mailing or shipping.
- Ensure all specimens have the correct name, date of birth, and date of collection before shipping. Incorrect or incomplete demographic information can lead to testing delays or rejection.
- Assign a staff person to double check all NBS specimens. Check for spot quality and all demographic information is complete before shipment.
- Ship to the correct address:
For Overnight/Courier Shipping (UPS, FedEx, etc):
Texas Department of State Health Services
Laboratory Services Section, MC 1947
1100 W. 49th Street
Austin, TX 78756-3199
For USPS Regular and Priority Mail:
Texas Department of State Health Services
Laboratory Services Section, MC 1947
PO Box 149341
Austin, TX 78714-9341
Need help?
Contact DSHS Newborn Screening Laboratory:
Phone: 1-888-963-7111 ext. 7585 or for local calls: 512-776-7585
Email Newborn Screening: NewbornScreeningLab@dshs.texas.gov
Posted December 9, 2024
DSHS Austin Newborn Screening Laboratory Thanksgiving Holiday Closure Reminder
On Thursday, November 28, 2024, the Texas Department of State Health Services (DSHS) Austin Newborn Screening (NBS) Laboratory will not be open, and no testing will occur. Please see below for more details:
- The NBS Laboratory will resume testing on Friday, November 29, 2024.
- DO NOT delay collection or shipment of NBS specimens.
- Ship dried specimens within 24 hours of collection. If mail or courier services are unavailable, ship as soon as possible.
- DSHS recommends shipping specimens using an overnight or trackable service like USPS Priority Mail, FedEx, or UPS.
- Per the FedEx website, FedEx will not be open on Thursday, 11/28/2024. FedEx will operate on a modified schedule November 27, 29, and 30th. Please plan accordingly for your site.
Newborn Screening Supply Order Requests
- Standard orders received before noon Wednesday, 11/27/2024 will get processed and shipped via FedEx Ground.
- Priority overnight orders received on Wednesday, 11/27/2024 will get processed and shipped via FedEx Priority Overnight.
- Orders received after noon Wednesday, 11/27/2024 through Sunday, 12/01/2024 will get filled Monday, 12/02/2024.
Need help?
Contact DSHS Newborn Screening Laboratory:
Phone: 1-888- 963-7111 ext. 7585 or for local calls: 512-776-7585
Email Newborn Screening: NewbornScreeningLab@dshs.texas.gov
Container Preparation: ContainerPrepGroup@dshs.texas.gov
Posted November 21, 2024
Format of Newborn Screening Results Reports Has Changed
On the afternoon of November 4, 2024, the Texas Department of State Health Services Laboratory implemented format changes to the newborn screening result report statements. Going forward, all results will be in the new format.
A summary of the format changes is available in a previous notice. That notice is accessible here: Newborn Screening Notices | Texas DSHS
Need help?
Contact DSHS Newborn Screening Laboratory:
Phone: 1-888- 963-7111 ext. 7585 or Locally: 512-776-7585
Email Newborn Screening: NewbornScreeningLab@dshs.texas.gov
Posted November 14, 2024
Changes to the Newborn Screening Result Reports
In early November, the Texas Department of State Health Services (DSHS) Laboratory will change the Newborn Screening (NBS) Result Reports. Examples of the updated NBS Result Reports are available for your review. Listed below is a summary of the changes to the report format.
Patient Information Changes:
- Condensed patient demographic and specimen information into three columns.
- Include some shortened field names to save space.
- No longer include the following fields:
- Mother’s Address
- Race/Ethnicity
- Test [i.e., 1st Test (under 7 days), 2nd Test (7 days or over)]
Result Information Changes:
- No longer display analyte and analyte result columns in the result table.
- However, analyte results will remain in the screening result notes for abnormal screens.
Testing Information Changes:
- No longer include a list of each NBS disorder screened at the bottom of the result report.
- Incorporate a web address and QR code that links to the NBS Disorders Screened webpage on the DSHS Laboratory NBS website.
Need help?
Contact DSHS Newborn Screening Laboratory:
Phone: 1-888-963-7111 ext. 7585 or Locally: 512-776-7585
Email Newborn Screening: NewbornScreeningLab@dshs.texas.gov
Posted November 04, 2024
Meet the Heroes Behind Newborn Screening: Celebrating the Teams Who Safeguard Texas Babies’ Health
As we celebrate Newborn Screening Awareness Month, we want to take this opportunity to introduce you to the dedicated testing teams behind the scenes, working tirelessly to protect the health of Texas newborns. These teams play a crucial role in early detection, helping save lives and ensuring that babies have the best possible start. Visit our NBS Awareness Website to learn more about their important work.
Here’s a look at our amazing teams:
- Biotinidase Deficiency Screening Team: Using color-changing reactions, this team tests for biotinidase deficiency, ensuring that babies who cannot process biotin get the treatment they need to avoid serious health issues.
- Enzyme Immunoassay (EIA) Team: This team screens for critical disorders like galactosemia, congenital hypothyroidism, congenital adrenal hyperplasia, and cystic fibrosis using enzyme-linked immunosorbent assays (ELISA). Their early detection efforts are vital in preventing life-threatening complications.
- Hemoglobinopathy Team: Working with isoelectric focusing and high-performance liquid chromatography techniques, this team detects genetic blood disorders like sickle cell disease and other hemoglobin variants, helping to ensure early intervention for affected infants.
- Severe Combined Immunodeficiency (SCID) and Spinal Muscular Atrophy (SMA) Team: This team screens for both SCID and SMA using advanced real-time polymerase chain reaction methods, enabling early diagnosis and treatment of these life-threatening conditions.
- Mass Spectrometry and LC Mass Spectrometry Teams: With its expertise in screening for x-linked adrenoleukodystrophy and over 40 metabolic conditions, including amino acid, fatty acid oxidation, and organic acid disorders, these teams ensure the earliest possible detection of rare metabolic disorders.
- DNA Analysis Team: Performing molecular confirmatory testing for disorders like cystic fibrosis and galactosemia, this team ensures accurate screening results, helping healthcare providers make informed decisions for newborn care.
Every day, these teams actively commit to improving the health of Texas newborns and make a real difference. Thank you for supporting our mission and celebrating Newborn Screening Awareness Month with us! Let’s continue raising awareness and supporting newborn screening!
Need help?
Contact DSHS Newborn Screening Laboratory:
Phone: 1-888- 963-7111 ext. 7585 or Locally: 512-776-7585
Email Newborn Screening: NewbornScreeningLab@dshs.texas.gov
Posted September 18, 2024
Critical Congenital Heart Disease – We Are Newborn Screening
September is Newborn Screening Awareness Month
Critical Congenital Heart Disease (CCHD) screening is a simple, non-invasive test designed to detect serious heart defects in newborns. CCHD is one of the leading causes of death in infants less than one-year-old. The Centers for Disease Control and Prevention (CDC) reports that about one in four babies born with a heart defect has a critical congenital heart defect. The Texas Health and Safety Code requires reporting of confirmed CCHD cases to the Texas Department of State Health Services.
The Special Projects Team coordinates CCHD duties. Visit We Are Newborn Screening | Texas DSHS to learn more about the specific roles these team members play in our CCHD activities.
For Questions Please Contact:
Newborn Screening Clinical Care Coordination
Phone: 512-776-3957
Toll-free: 800-252-8023 ext. 3957
Newborn@dshs.texas.gov
Posted September 16, 2024
We Are Newborn Screening – Meet the teams that keep us moving.
We would like to introduce you to the teams that form the backbone of the preanalytical process in Newborn Screening (NBS). Each of these teams work to ensure the NBS process gets off to a good start. Accurate and timely screening for newborns begins with the following teams:
- Lab Supply/Container Preparation Teams: maintain the availability of essential supplies needed for specimen collection and testing.
- NBS Check-In Team: manages the initial receipt and verification of all specimens.
- Specimen Logistics Team: ensures the smooth flow of specimens from collection to testing.
- Specimen Preparation: prepares the specimens for testing. Processes specimens into testing plates ensuring reliable NBS results.
We invite you to learn more about each team by visiting the 2024 NBS Awareness Month Webpage. You can see pictures of our amazing staff and their work areas.
For Questions Please Contact:
Newborn Screening Laboratory Educators:
Phone: 512-776-7585
Toll-free: 1-888-963-7111, ext. 7585
Fax: 512-776-7157
NewbornScreeningLab@dshs.texas.gov
Posted September 11, 2024
Texas Early Hearing Detection and Intervention (TEHDI) – We Are Newborn Screening
September is Newborn Screening Awareness Month
Did you know early intervention for hearing loss can make a world of a difference in a child’s development? The TEDHI Team promotes understanding of critical timelines and steps to manage hearing loss. Each year in Texas, about 500 babies are born deaf or hard of hearing. Hearing loss is the second most common birth defect in Texas.
We encourage you to take a moment to meet the team and learn about all the work they do to help Texas children. Together we can continue to support children’s development and well-being.
For Questions Please Contact:
TEHDI, Newborn Screening
Phone: 512-483-7417
Toll-free: 800-252-8023, ext. 6616
TEDHI@dshs.texas.gov
Posted September 9, 2024
Newborn Screening Guidance During Hurricane Season
As you prepare for possible bad weather, please remember:
- Do Not Delay Newborn Screening (NBS) Specimen Collection.
- Collect 1st screens at 24-48 hours of age.
- Collect 2nd screens between 7-14 days of age, a minimum of 168 hours.
- Ensure the parent/guardian contact information will be valid throughout any potential relocation.
- For patient transfers - Include newborn screening specimen collection status in transfer documentation. Notify receiving facility of specimen collection need if not collected before transfer.
- Allow NBS specimens to dry a minimum of 3 hours.
- DO NOT put specimens in air-tight sealed containers.
- Ship As Soon As Possible.
- If you use courier services, contact the courier for extra information about your location.
- During courier delays store dried specimens at room temperature in a dry location.
- Walk your specimens inside the postal location. Do not drop specimens in the blue mailboxes outside. Mailboxes become very hot, humid and can flood during high water events.
Ensuring samples reach the lab in good condition helps provide accurate screening test results.
Need help?
Contact DSHS Newborn Screening Laboratory:
Phone: 1-888- 963-7111 ext. 7585 or Locally: 512-776-7585
Email Newborn Screening: NewbornScreeningLab@dshs.texas.gov
Posted September 9, 2024
Together – We Are Newborn Screening September is Newborn Screening Awareness Month
It takes a team effort to manage a newborn screening (NBS) program as big as Texas. Together,
since 1965 we have made a difference in the lives of our youngest Texans. Texas is home to the
largest NBS program in the United States. This year, we are shining a spotlight on the incredible
teams working behind the scenes to ensure every baby born in Texas gets the care they need.
In 2023, we achieved remarkable milestones:
- Screened over 760,000 specimens from almost 400,000 babies for 55 disorders.
- Provided hearing screening for babies born in Texas.
- Screened newborns for critical congenital heart disease.
These accomplishments are a testament to the dedication and hard work of our teams. We
encourage you to take a moment to recognize your team and all the work they put into
making NBS successful in Texas. Together with our partners, we operate a Texas-sized NBS
program that gives babies the best possible start to a healthy life. Let’s continue to work
together to protect and support our youngest Texans.
For Questions Please Contact:
Newborn Screening Laboratory Educators:
Phone: 512-776-7585
Toll-free: 1-888-963-7111, ext. 7585
Fax: 512-776-7157
NewbornScreeningLab@dshs.texas.gov
Posted September 4, 2024
Newborn Screening Quality Improvement Hints Caked, Clotted, or Layered Blood on Filter Paper
In 2023, over 2,102 newborn screening specimens were rejected because the blood was caked, clotted, or layered onto the filter paper. All these specimens required a recollection and caused critical delays in testing.
Why is caked, clotted, or layered specimens rejected?
The supplied newborn screening filter paper can hold a specific amount of blood. Caked, clotted, or layered blood specimens have too much blood. Specimens with too much blood can cause inaccurate results. These specimens will be unsatisfactory for testing.
Download a copy of the Quality Improvement Hint: Blood was caked, clotted, or layered.
View other NBS Healthcare Provider Resources.
Questions?
1-888-963-7111 ext. 7333 or locally 512-776-7333
NewbornScreeningLab@dshs.texas.gov
Posted August 15, 2024
Changes to 2024 Newborn Screening Specimen Collection Kits
2024 newborn screening specimen collection kits will begin shipping soon. The new collection kits begin with serial numbers starting 24 (24XXXXXXX). The following are changes from previous kits.

Please ensure all information is complete, accurate, and legible before shipping specimens to DSHS. Incorrect or missing information can cause:
- specimen rejection
- delayed testing
- incorrect test results and reports
Are older versions of the collection kit still acceptable?
Yes, all collection kits that have not expired are still acceptable. The expiration date is printed on all collection kits.
For Questions Please Contact:
Newborn Screening Laboratory
Phone: 512-776-7333
Toll-free: 1-888-963-7111 ext. 7333
Email Newborn Screening: NewbornScreeningLab@dshs.texas.gov
Posted August 12, 2024
Newborn Screening Guidance During Hurricane Season
As you prepare for possible bad weather, please remember:
- Do Not Delay Newborn Screening (NBS) Specimen Collection.
- Collect 1st screens at 24-48 hours of age.
- Collect 2nd screens between 7-14 days of age, a minimum of 168 hours.
- Ensure the parent/guardian contact information will be valid throughout any potential relocation.
- For patient transfers - Include newborn screening specimen collection status in transfer documentation. Notify receiving facility of specimen collection need if not collected before transfer.
- Allow NBS specimens to dry a minimum of 3 hours.
- DO NOT put specimens in air-tight sealed containers.
- Ship As Soon As Possible.
- If you use courier services, contact the courier for extra information about your location.
- During courier delays store dried specimens at room temperature in a dry location.
- Walk your specimens inside the postal location. Do not drop specimens in the blue mailboxes outside. Mailboxes become very hot, humid and can flood during high water events.
Ensuring samples reach the lab in good condition helps provide accurate screening test results.
Need help?
Contact DSHS Newborn Screening Laboratory:
Phone: 1-888- 963-7111 ext. 7585 or Locally: 512-776-7585
Email Newborn Screening: NewbornScreeningLab@dshs.texas.gov
Posted July 3, 2024
Texas Summer Heat Can Affect Specimen Quality
Shipping newborn screening (NBS) specimens during summer months can be a challenge. Heat and humidity can affect the stability of the specimens, causing inaccurate results. It is important to transport specimens at a controlled temperature, if possible.
Here are some key points to consider:
- Schedule mail pickups inside your location. Do not place specimens on outside of facility door for pickup.
- Walk your specimens inside the postal location. Do not drop specimens in the blue mailboxes outside. Mailboxes become very hot and humid.
- Never leave specimens inside a non-running closed automobile.
Ensuring samples reach the lab in good condition help provide accurate screening test results.
Need help?
Contact DSHS Newborn Screening Laboratory:
Phone: 1-888- 963-7111 ext. 7585 or Locally: 512-776-7585
Email Newborn Screening: NewbornScreeningLab@dshs.texas.gov
Posted June 7, 2024
Correct Website to Parent Decision Form for Storage and Use of Newborn Screening Blood Spot Cards Information
The Texas Department of State Health Services (DSHS) migrated its website to a new platform in 2023. The move deactivated a website address listed on all the Newborn Screening (NBS) collection kits currently in use. The Parent Decision Form for Storage and Use of Newborn Screening Blood Spot Cards page now contains the wrong website address. See the image below “for questions or more information” section. The affected kits are those with serial numbers starting with 21-XXXXXXX, 22-XXXXXXX, and 23-XXXXXXX.
The correct website address to direct parents to Newborn Screening - Frequently Asked Questions.
This summer, we will begin distributing the 2024 NBS collection kits (24-XXXXXXX) with the correct website address.

For Questions Please Contact:
Newborn Screening Laboratory
Phone: 512-776-7333
Toll-free: 1-888-963-7111 ext. 7333
Email Newborn Screening: NewbornScreeningLab@dshs.texas.gov
Posted May 20, 2024
Newborn Screening Quality Improvement Hints Importance of Birthweight
DSHS Newborn Screening (NBS) Laboratory evaluates test results based on birthweight. Leaving the birthweight blank on the NBS demographic page may delay testing. Test results may not be accurate if the weight is wrong or not in grams.
Download a copy of the Quality Improvement Hint: Importance of Birthweight
View a weight conversion chart and other NBS Healthcare Provider Resources.
Questions?
1-888-963-7111 ext. 7333 or locally 512-776-7333
NewbornScreeningLab@dshs.texas.gov
Posted March 22, 2024
Newborn Screening Laboratory Addresses for Receiving Specimens
The Texas Department of State Health Services (DSHS) Laboratory can receive both overnight shipments and regular United States Postal Service (USPS) mail deliveries. Submitters should be mindful of the time between collection, shipment, and delivery. DSHS recommends shipping specimens using an overnight or trackable service like USPS Priority Mail, FedEx, or UPS.
The clock starts ticking the day of collection, which equals day one. Newborn screening (NBS) specimens are unsatisfactory if received after 13 days of collection and will need a recollection. Best practice is to allow NBS specimens to dry a minimum of 3 hours, per Clinical and Laboratory Standards Institute (CLSI). Ship specimen same day using an overnight or trackable service.
For Overnight/Courier Shipping (UPS, FedEx, etc.):
Texas Department of State Health Services
Laboratory Services Section, MC 1947
1100 W. 49th Street
Austin, TX 78756-3199
For USPS Regular and Priority Mail:
Texas Department of State Health Services
Laboratory Services Section, MC 1947
PO Box 149341
Austin, TX 78714-9341
Need help?
Contact DSHS Newborn Screening Laboratory:
Phone: 1-888- 963-7111 ext. 7585 or Locally: 512-776-7585
Email Newborn Screening: NewbornScreeningLab@dshs.texas.gov
Posted March 4, 2024
Clarification: Changes to Newborn Screening Cystic Fibrosis Testing and Result Reporting Statements Effective February 5, 2024
The Texas DSHS Newborn Screening (NBS) Laboratory updated and added new result codes for Cystic Fibrosis to improve fidelity in reporting by reducing the frequency of manual result entry. No changes were made to the DSHS CF DNA variant panel. For all existing CF DNA results, the CFTR mutation analyte result in the result grid of the report was updated to match the language in the result notes (variant instead of mutation per ACMG guidelines). Eighty new CFDNA result combinations that previously required free text entry were added to the list of results linked below. These updates went into effect on February 5, 2024.
View a full list of all Newborn Screening Laboratory Results.
Reminder: Review the result screening note(s) for all CF DNA results.
Questions?
Contact DSHS Newborn Screening Laboratory:
Phone: 1-888- 963-7111 ext. 7585 or Locally: 512-776-7585
Email Newborn Screening: NewbornScreeningLab@dshs.texas.gov
Posted February 27, 2024
Changes to Newborn Screening Cystic Fibrosis Testing and Result Reporting Statements Effective February 5, 2024
The Texas DSHS Newborn Screening (NBS) Laboratory updated and added new result codes for Cystic Fibrosis to improve fidelity in reporting by reducing the frequency of manual result entry. For all existing CF DNA results, the CFTR mutation analyte result in the result grid of the report was updated to match the language in the result notes (variant instead of mutation per ACMG guidelines). Additionally, 80 new CF DNA results were added. These updates went into effect on February 5, 2024.
View a full list of all Newborn Screening Laboratory Results.
Reminder: Review the result screening note(s) for all CF DNA results.
Questions?
Contact DSHS Newborn Screening Laboratory:
Phone: 1-888- 963-7111 ext. 7585 or Locally: 512-776-7585
Email Newborn Screening: NewbornScreeningLab@dshs.texas.gov
Posted February 20, 2024
Cost of the DSHS Newborn Screening Kit will Increase January 1, 2024
Effective January 1, 2024, the cost of Private Pay Insurance/Self-Pay newborn screening (NBS) kits will increase by 8%, from $63.55 to $68.63. Insurance/Self-Pay NBS kits ordered on or after January 1, 2024, will be billed at the new price of $68.63.
The purpose of this increase will be to support increased costs for operations and is not for the addition of any new conditions to the Department of State Health Services newborn Screening panel.
View complete details, including the current Austin and South Texas DSHS Laboratory fee schedules.
For Questions Please Contact:
Newborn Screening Laboratory
Phone: 512-776-7333
Toll-free: 1-888-963-7111 ext. 7333
NewbornScreeningLab@dshs.texas.gov
Posted December 13, 2023
DSHS Newborn Screening Improvement to Primary Congenital Hypothyroidism Testing Methodology
The Department of State Health Services (DSHS) Newborn Screening (NBS) Program—in collaboration with the CDC, Texas endocrinology consultants, and national experts—conducted an improvement project to optimize screening for Primary Congenital Hypothyroidism (CH).
Goals of the project:
- Optimize screening algorithm for identification of CH and reduce the false positive rate and age at diagnosis.
- Reduce the time to provider notification for critically high TSH levels.
- Improve outcomes and reduce costs to the overall healthcare system.
On December 4, 2023, changes will be implemented to the initial screening CH testing methodology based on this project’s findings.
- Specimens collected before 7 days of life will be screened using Thyroid Stimulating Hormone (TSH). A subset of specimens with slightly elevated TSH levels will be retested for both TSH and Thyroxine (T4) levels.
- All specimens collected at 7 days of life or later will be screened using both Thyroxine (T4) and Thyroid Stimulating Hormone (TSH).
There will be no changes to result reporting statements, HL7 reporting or recommended follow-up activities at this time.
Where can I find more information on Primary Congenital Hypothyroidism (CH)?
Find more information at Congenital Hypothyroidism ACT sheet.
For Questions Please Contact:
Newborn Screening Laboratory
Phone: 512-776-7333
Toll-free: 1-888-963-7111 ext. 7333
NewbornScreeningLab@dshs.texas.gov
Posted December 04, 2023
